![]()
 Fisher and Paykel Healthcare Reusable Breathing Circuit: Class I Recall – Pinholes in Tubes Fisher and Paykel Healthcare Reusable Breathing Circuit: Class I Recall – Pinholes in TubesFDA notified healthcare professionals that the Fisher & Paykel Healthcare Reusable Breathing Circuit, Model 900MR068, was recalled due to pinholes in the tubes used in the reusable breathing circuit. If these pinholes are not detected during the standard leak test before patient use, it could potentially result in a gas leak in the breathing system, which may lead to a loss of pressure for the intended ventilation therapy. Usage of the defective device may result in patient death. More information |
 Medication Guides for Certain Prescription Medicines Medication Guides for Certain Prescription MedicinesThe FDA wants you to know the importance of reading Medication Guides—paper handouts that come with many prescription medicines. “The information in a Medication Guide is very important when it comes to getting the most benefit for your health from a medicine,” says Paul Seligman, M.D., M.P.H., former Associate Director for Safety Policy and Communication in FDA’s Center for Drug Evaluation and Research. “These guides address issues that are specific to particular drugs and drug classes, and they contain FDA-approved information that can help patients avoid serious adverse events. To continue reading this Consumer Update or for more information about currently FDA approved Medication Guides.
|
Descargo de responsabilidad: La FDA reconoce la necesidad de proporcionar información sobre seguridad importante en idiomas distintos al inglés. Hacemos lo mejor posible para proporcionar versiones en español precisas y oportunas de nuestras Comunicaciones de Seguridad de Medicamentos. Sin embargo, en caso que existiera discrepancias entre las versiones en ingles y en español, la información contenida en la versión en inglés es la que se considera como versión oficial. Si tiene alguna pregunta, por favor contáctese con Division of Drug Information en druginfo@fda.hhs.gov. Comunicaciones de la FDA |
 For more important safety information on human drug and devices or to report a serious problem, please visit MedWatch. For more important safety information on human drug and devices or to report a serious problem, please visit MedWatch.
|
![]()
 FDA recognizes the significant public health consequences that can result from drug shortages and takes tremendous efforts within its legal authority to address and prevent drug shortages. These shortages occur for many reasons, including manufacturing and quality problems, delays, and discontinuations. When quality/manufacturing and quality problems, delays, and discontinuations. When quality/manufacturing issues are discovered by the company or the public and reported to FDA or are found by FDA upon inspection, the FDA works closely with the firm to address risks involved to prevent harm to patients. FDA also considers the impact a shortage would have on patient care and access and works with the firm to restore supplies while also ensuring safety for patients. FDA recognizes the significant public health consequences that can result from drug shortages and takes tremendous efforts within its legal authority to address and prevent drug shortages. These shortages occur for many reasons, including manufacturing and quality problems, delays, and discontinuations. When quality/manufacturing and quality problems, delays, and discontinuations. When quality/manufacturing issues are discovered by the company or the public and reported to FDA or are found by FDA upon inspection, the FDA works closely with the firm to address risks involved to prevent harm to patients. FDA also considers the impact a shortage would have on patient care and access and works with the firm to restore supplies while also ensuring safety for patients.More information
|
Drug Shortages Voluntarily Reported by Manufacturers During the Past 2 Weeks:
- No current shortages reported
Drug Shortages Resolved During the Past 2 Weeks:
- No current shortages resolved
Drugs to be Discontinued Announced During Past 2 Weeks:
- Chloral Hydrate (Somnote) soft gel capsule
|
![]()
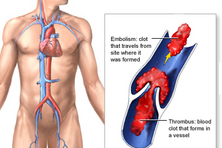 | Eliquis approved to reduce the risk of stroke, blood clots in patients with non-valvular atrial fibrillation FDA approved the anti-clotting drug Eliquis (apixaban), an oral tablet used to reduce the risk of stroke and dangerous blood clots (systemic embolism) in patients with atrial fibrillation that is not caused by a heart valve problem. More information |
 Sirturo approved to treat multi-drug resistant pulmonary tuberculosis Sirturo approved to treat multi-drug resistant pulmonary tuberculosisFDA approved Sirturo (bedaquiline) as part of combination therapy to treat adults with multi-drug resistant pulmonary tuberculosis (TB) when other alternatives are not available. TB is an infection caused by Mycobacterium tuberculosis and is one of the world’s deadliest diseases. It is spread from person to person through the air and usually affects the lungs, but it can also affect other parts of the body such as the brain and kidneys. More information |
Fulyzag approved as first anti-diarrheal drug for HIV/AIDS patients
FDA approved Fulyzaq (crofelemer) to relieve symptoms of diarrhea in HIV/AIDS patients taking antiretroviral therapy, a combination of medicines used to treat HIV infection. More information
 For information on drug approvals or to view prescribing information and patient information, please visit Drugs@FDA or DailyMed. For information on drug approvals or to view prescribing information and patient information, please visit Drugs@FDA or DailyMed. |
![]()
 Guidance Documents on MDUFA III Guidance Documents on MDUFA IIIThe Food and Drug Administration Safety and Innovation Act (Public Law 112-144) includes the Medical Device User Fee Amendments of 2012, or MDUFA III. MDUFA III will take effect on October 1, 2012 and will sunset in five years on October 1, 2017. Device user fees were first established by Congress in 2002. Medical device companies pay fees to FDA when they register their establishment and list their devices with the agency, whenever they submit an application or a notification to market a new medical device in the U.S. and for certain other types of submissions. The FDA are issuing a series of guidance documents that help explain key provisions of MDUFA III. These guidance documents include 3 new guidance’s dated December 31, 2012. Request for Comments: Framework for Pharmacy Compounding – State and Federal Roles
Request for Comments: Custom Devices
|
 Request for Comments: Draft Guidance for Industry – Electronic Source Data in Clinical Investigations
Request for Comments: Draft Guidance for IRBs, Clinical Investigators, and Sponsors – IRB Responsibilities for Reviewing the Qualifications of Investigators, Adequacy of Research Sites, and the Determination of Whether an IND/IDE Is Needed
Draft Guidances for Industry: Safety Considerations for Product Design to Minimize Medication Errors
Request for Comments and Information on Initiating a Risk Assessment for Establishing Food Allergen Thresholds Request for Comments: Draft Guidance for Industry on Enrichment Strategies for Clinical Trials to Support Approval of Human Drugs and Biological Products |
 Request for Comments: Information To Accompany Humanitarian Device Exemption Applications and Annual Distribution Number Reporting Requirements Request for Comments: Information To Accompany Humanitarian Device Exemption Applications and Annual Distribution Number Reporting RequirementsFDA invites comments on these topics: (1) Whether the proposed collection of information is necessary for the proper performance of FDA’s functions, including whether the information will have practical utility; (2) the accuracy of FDA’s estimate of the burden of the proposed collection of information, including the validity of the methodology and assumptions used; (3) ways to enhance the quality, utility, and clarity of the information to be collected; and (4) ways to minimize the burden of the collection of information on respondents, including through the use of automated collection techniques, when appropriate, and other forms of information technology. Comments are due by February 15, 2013. More information
|
Request for Comments Date Extended: Information on Nicotine Replacement Therapies and Smoking-Cessation Products
FDA is extending the comment period for the notice of public hearing that appeared in the Federal Register of November 28, 2012 (77 FR 70955). In the public hearing notice, FDA requested comments on FDA consideration of applicable approval mechanisms and additional indications for nicotine replacement therapies (NRTs), and input on a report to Congress examining the regulation and development of innovative products and treatments for tobacco dependence. The Agency is taking this action to allow interested persons additional time to submit comments. More information
 Request for Comments: Draft Guidance for Industry – Development of Vaginal Microbicides for the Prevention of Human Immunodeficiency Virus Infection
Request for Comments: Clinical Development Programs for Disease-Modifying Agents for Peripheral Neuropathy and Public Workshop
Request for Comments: Draft Guidance for Industry and FDA Staff – Design Considerations for Devices Intended for Home Use Request for Comments: Impact of Approved Drug Labeling on Chronic Opioid Therapy |
![]()
 Drug Residue in our Food Supply Drug Residue in our Food SupplyIllegal drug residues in the nation’s food supply are a concern to the Food and Drug Administration. The Center for Veterinary Medicine’s Division of Compliance is responsible for reviewing violative residues reported to the Agency by the USDA’s Food Safety and Inspection Service. The residues are ranked using a Risk Model to ensure Federal and/or State investigation of the residues of greatest public health concern for which assignments are issued. The Drug Residue Compliance Team provides regulatory support and outreach to prevent illegal drug residues by reviewing inspectional evidence sent to us by the FDA District Offices. More information |
| FDA Provides a Portal to India |  |
 FDA proposes new food safety standards for foodborne illness prevention and produce safety FDA proposes new food safety standards for foodborne illness prevention and produce safetyThe U.S. Food and Drug Administration today proposed two new food safety rules that will help prevent foodborne illness. The proposed rules implement the landmark, bipartisan FDA Food Safety Modernization Act (FSMA) and are available for public comment for the next 120 days. The FDA encourages Americans to review and comment on these important proposed rules. More information New Drug Attacks Resistant TB by Margaret A. Hamburg, M.D., Commissioner of the U.S. Food and Drug Administration
|
| FDA Commemorates 30th Anniversary of the Orphan Drug Act By Gayatri R. Rao, M.D., J.D., Director for The Office of Orphan Products Development When President Reagan signed the Orphan Drug Act 30 years ago, he enacted a critically important piece of health care legislation. The passage of this Act on January 4, 1983, was monumental because it created—for the first time—incentives to develop desperately needed medical products for Americans suffering with rare diseases. Until that point, development of such products was very limited. For instance, in the decade leading up to the passage of the Orphan Drug Act, only 10 industry-supported products for rare diseases were brought to market. To continue reading this blog, see FDA Voice Blog, January 7, 2013 |  |
Basing Food Safety Standards on Science and Prevention By Margaret Hamburg, M.D., Commissioner of the U.S. Food and Drug Administration
Two of my highest priorities as FDA commissioner have been strengthening the scientific foundation of FDA’s regulatory decisions and ensuring the safety of an increasingly complex and global food supply. That’s why I take such pride in FDA’s proposal of two rules that set science-based standards for the prevention of foodborne illnesses. One will govern facilities that produce food, and the other concerns the safety of produce. The Preventive Controls for Human Food rule proposes that food companies—whether they manufacture, process, pack or store food— put in place controls to minimize and reduce the risk of contamination. The Produce Safety rule proposes that farms that grow, harvest, pack or hold fruits and vegetables follow standards aimed at preventing their contamination. To continue reading this blog, see FDA Voice Blog, January 8, 2013
Cord Blood Banking – Information for Consumers
Expecting a baby can be a very exciting time for soon-to-be-parents. It can also be very confusing, with many decisions to make. One choice prospective parents often face is whether to donate, bank or discard their baby’s cord blood. Did you know that the Food and Drug Administration (FDA) regulates cord blood? Here is some information for expectant parents about the regulations in place designed to help ensure the safety of cord blood for transplantation. To continue reading this information for consumers
![]()
FDA advisory committee meetings are free and open to the public. No prior registration is required to attend. Interested persons may present data, information, or views, orally at the meeting, or in writing, on issues pending before the committee.
Other types of meetings listed may require prior registration and fees. Click on “More information” for details about each meeting.
![]()
 Endocrinologic and Metabolic Drugs Advisory Committee Meeting Endocrinologic and Metabolic Drugs Advisory Committee MeetingDate: January 10, 2013 The committee will discuss new drug application (NDA) 204042, canagliflozin tablets, proposed trade name INVOKANA, submitted by Janssen Research and Development, LLC. Canagliflozin is a member of the sodium-glucose co-transporter 2 (SGLT2) inhibitors, and was developed as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. More information |
Cellular, Tissue and Gene Therapies Advisory Committee Meeting
Date: January 15, 2013
On January 15, 2013 from 2 p.m. to approximately 4:30 p.m., the Committee will meet, by teleconference, in open session, to hear updates on the research programs in the Laboratory of Immunology, Division of Therapeutic Proteins, Office of Biotechnology Products, Center for Drug Evaluation and Research, FDA. From approximately 4:30 p.m. to 5:30 p.m., the meeting will be closed to permit discussion where disclosure would constitute a clearly unwarranted invasion of personal privacy (5 U.S.C. 552b(c)(6)). The committee will discuss reports of the intramural research programs and make recommendations regarding personnel staffing decisions. More information
Drug Safety and Risk Management Advisory Committee Meeting
Dates: January 24-25, 2013
The committee will discuss the public health benefits and risks, including the potential for abuse, of drugs containing hydrocodone either combined with other analgesics or as an antitussive. The Department of Health and Human Services received a request from the Drug Enforcement Administration for a scientific and medical evaluation and scheduling recommendation for these products in response to continued reports of misuse, abuse, and addiction related to these products. The committee will also discuss the impact of rescheduling these hydrocodone products from Schedule III to Schedule II. More information
 Pulmonary-Allergy Drugs Advisory Committee Meeting Pulmonary-Allergy Drugs Advisory Committee MeetingDate: January 29, 2013 The committee will discuss the new drug application (NDA) 203108, for olodaterol (proposed trade name Striverdi Respimat) metered dose inhaler, sponsored by Boehringer Ingelheim, for the proposed indication of long-term, once-daily maintenance bronchodilator treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD), including chronic bronchitis and/or emphysema. More information
Pulmonary-Allergy Drugs Advisory Committee Meeting |
![]()
Public Hearing: Impact of Approved Drug Labeling on Chronic Opioid Therapy
Dates: February 7-8, 2013
Over the past several years, the role of opioid drugs in treating chronic pain has been an increasingly common subject of public discussion. FDA and other policymakers have been at the forefront of these debates, striving to find a balance between minimizing opioid drug abuse and misuse, while simultaneously enabling appropriate access to pain-relieving drugs. The July 9, 2012, approval of the Risk Evaluation Management Strategy for extended-release (ER) and long-acting (LA) opioid analgesics is a recent example of FDA’s ongoing commitment to ensuring that the benefits of these types of opioid drugs continue to outweigh their risks. More information
Public Workshop: Clinical Development Programs for Disease-Modifying Agents for Peripheral Neuropathy
Dates: February 11-12, 2013
FDA is announcing this public workshop to further the understanding of the development of disease-modifying agents for the treatment of painful peripheral neuropathies. Discussion will focus on possible therapeutic targets for these agents, the types of painful peripheral neuropathies amenable to treatment with disease-modifying agents, and clinical trial design. More information
Risk Communication Advisory Committee Meeting
Date: February 12, 2013
The committee will discuss general factors in risk communication about FDA regulated products, including approaches to avoid message fatigue and related communication barriers such as prevention or warning fatigue or inaccurate risk perception. More information
 Medical Imaging Drugs Advisory Committee Meeting Medical Imaging Drugs Advisory Committee MeetingDate: February 14, 2013 The committee will discuss new drug application (NDA) 204781, proposed trade name DOTAREM (gadoterate meglumine injection), application submitted by Guerbet, LLC. The proposed indication (use) for this product is for magnetic resonance imaging in brain (intracranial), spine, and associated tissues in adults and pediatric patients (from neonates to 17 years of age) to detect and visualize areas with disruption of the blood brain barrier (specialized tissues that help protect the brain) and/or abnormal vascularity (abnormal blood circulation). More information
|
Neurological Devices Panel of the Medical Devices Advisory Committee Meeting
Date: February 22, 2013
The committee will discuss, make recommendations, and vote on information regarding the premarket approval application (PMA) for the NeuroPace RNS System sponsored by NeuroPace, Inc. The RNS System is indicated for use as an adjunctive therapy in reducing the frequency of seizures in individuals 18 years of age or older with partial onset seizures from no more than two foci that are refractory to two or more antiepileptic medications. More information
![]()
Reproductive Health Drugs Advisory Committee Meeting
Date: March 4, 2013
The morning session, the committee will discuss new drug application (NDA) 022506, gabapentin 600 milligram (mg) tablets, submitted by Depomed, Inc., for the proposed indication of treatment of moderate to severe vasomotor symptoms due to menopause. During the afternoon session, the committee will discuss NDA 204516, paroxetine mesylate 7.5 mg capsules, submitted by Noven Therapeutics, LLC, for the proposed indication of treatment of moderate to severe vasomotor symptoms associated with menopause. More Information
Pulmonary-Allergy Drugs Advisory Committee Meeting
Date: March 7, 2013
The committee will discuss the new drug application (NDA) 204275 for fluticasone furoate and vilanterol dry powder inhaler (proposed tradename BREO ELLIPTA), sponsored by GlaxoSmithKline, for the long-term maintenance treatment of airflow obstruction and for reducing exacerbations in patients with chronic obstructive pulmonary disease. More information
![]()
Orthopaedic and Rehabilitation Devices Panel of the Medical Devices Advisory Committee Meeting
Date: April 5, 2013
The committee will discuss and make recommendations regarding the possible reclassification of Shortwave Diathermy devices. On July 6, 2012 (77 FR 39953), FDA issued a proposed rule which, if made final, would make Shortwave Diathermy devices Class III, requiring premarket approval. In response to the proposed rule, FDA received petitions under section 515(b)(2)(B) of the Federal Food, Drug, and Cosmetic Act requesting a change in classification. The reclassification petitions are available for public review and comment at www.regulations.gov under docket number FDA-2012-N-0378.More information
Please visit FDA’s Advisory Committee page to obtain advisory committee meeting agendas, briefing materials, and meeting rosters prior to the meetings. You may also visit this page after meetings to obtain transcripts, presentations, and voting results. For additional information on other agency meetings please visit Meetings, Conferences, & Workshops. |
![]()
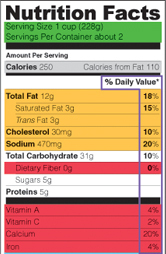 Nutrition Facts Label: 20 and Evolving Nutrition Facts Label: 20 and EvolvingWhen you’re walking down the aisles of a supermarket, it’s not unusual to see fellow shoppers reading the information on the back of a food package, box or can. They might want to know how many calories are in the food, or they might be watching their sodium intake.
They could be trying to limit sugars and eat more dietary fiber. Or they could be parents trying to make the most nutritious choices for their children.
All this information is available thanks to an important addition to food packaging that was introduced to the American public 20 years ago: the Nutrition Facts label. This familiar rectangular box provides, in a standard format, important information about the nutritional content for most packaged foods, including breads, cereals, canned and frozen foods, snacks, desserts and drinks. More information |
 FDA Strengthening our Food Safety Foundation FDA Strengthening our Food Safety FoundationFor the FDA, prevention is at the heart of food safety. “Preventing problems before they cause harm is not only common sense, it is the key to food safety in the 21st century,” says FDA Commissioner Margaret A. Hamburg, M.D. “We cannot afford to wait until people become ill to realize there is a problem.” Prevention is the core principle of the FDA Food Safety Modernization Act that President Obama signed into law in 2011, creating a blueprint for the most sweeping changes to the nation’s food protection system since Theodore Roosevelt held office. More information |
 Understanding Antidepressant Medications More Consumer Updates Artículos en Español |
![]()
 Device Use Safety Tips
FDA Basics
|
 Have a question about what the Center for Drug Evaluation and Research (CDER) does? Ask Janet Woodcock, M.D., Director, CDER, FDA Have a question about what the Center for Drug Evaluation and Research (CDER) does? Ask Janet Woodcock, M.D., Director, CDER, FDAFDA will select some of your questions to answer each month. Due to the volume of e-mails we receive, we won’t be able to answer each question in this format. We may edit your questions for brevity or clarity. More information
To read questions and answers, see MailBag. |

Animal Health Literacy CVM Pet Facts |
 Center for Food Safety and Applied Nutrition Center for Food Safety and Applied NutritionThe Center for Food Safety and Applied Nutrition, known as CFSAN, carries out the mission of the Food and Drug Administration (FDA). The Center provides services to consumers, domestic and foreign industry and other outside groups regarding field programs; agency administrative tasks; scientific analysis and support; and policy, planning and handling of critical issues related to food and cosmetics. More information
Food Facts for You |
 Tobacco Products Resources for You Tobacco Products Resources for YouFederal resources to help you quit using tobacco products and to help you learn more about youth tobacco prevention, effective treatment for nicotine addiction, and tobacco research and statistics. More information |
FDA News / Product Safety Info (1-9-2013)